 his week a
paper showed
up on ArXiv claiming to have discovered the first protein from outer space. It's
too early to say whether the protein, which they extracted from a meteorite, is biotic
in origin or whether it was formed chemically. The authors, Malcom McGeoch, Sergei
Dikler, and Julie McGeoch, named it hemolithin and tried to determine its structure
using MALDI mass spectrometry.
his week a
paper showed
up on ArXiv claiming to have discovered the first protein from outer space. It's
too early to say whether the protein, which they extracted from a meteorite, is biotic
in origin or whether it was formed chemically. The authors, Malcom McGeoch, Sergei
Dikler, and Julie McGeoch, named it hemolithin and tried to determine its structure
using MALDI mass spectrometry.
MALDI is a standard way of identifying proteins. You mix the protein with a chemical called a matrix, apply it on a metal plate, then blast it with a laser to vaporize it and measure the mass of the molecules that enter the mass spectrometer. MALDI is very good at seeing intact high molecular weight molecules—most other instruments max out at 3000 or so. But MALDI is not not very good at determining structure, it's not quantitative, and the lower mass part of the spectrum is mostly artifacts.
The authors say hemolithin consists of two linear chains of glycine and hydroxyglycine capped at either end by iron atoms bound to oxygen. They detected lithium by its atomic mass of 7 and proposed that it's bound as an adduct, which is to say non-covalently, to two of the oxygens at the ends of the molecule.
They took care to avoid contamination when extracting the protein, and they found enhanced deuterium (D), which is an accepted sign of extraterrestrial origin. Deuterium on a protein exchanges with hydrogen very rapidly whenever water is present, so the amount of D is probably underestimated, making it hard to say where it originated.
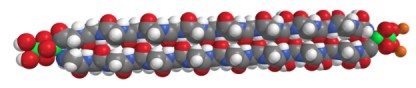
Space-filling model of hemolithin proposed by McGeoch et al. Red=O, Gray=C, Blue=N, Green=Fe, Orange=Li. Hemolithin is a very small protein containing two peptide strands of 2320 Da, each 16 amino acids long, for a total mass of 4641 Daltons. Some proteins of 15 and 17 amino acids were also detected.
By cranking up the laser intensity, they could fragment the protein into smaller chunks. This produced what people call a “ladder,” a discrete series of masses that's typical of a polymer. However, there were many artifactual masses caused by matrix components that had to be removed manually. Only three fragments were abundant enough to do MS/MS fingerprinting; they identified the remainder by matching their masses to the expected masses. For example, they found a mass at 401.086 m/z, which is reasonably close to 401.166. This is within 200 ppm of the expected mass of a glycine 7-mer plus two hydrogens.
I've seen many published papers where wildly incorrect structures were deduced this way, so it would have been nice to see the results confirmed by a conventional proteomic analysis. For an unknown protein like this one, they'd use a method called de novo sequencing, which would have told us whether there's any pattern in how the glycines and hydroxyglycines are organized. That might not be so easy, as there were probably very small amounts of the protein in that meteorite.
The iron complexation looks quite weird. Iron is octahedral and binds six ligands. In hemoglobin, four of the six bind to nitrogens on nearby histidines; the rest are used to bind dioxygen. By contrast, one of the irons in hemolithin is complexed to five oxygens, while the other is complexed to three oxygens, a nitrogen, and a carbon. This is a Fe-O3-Fe hematite motif which is not seen in Earth proteins and looks to me very unnatural and inorganic. But maybe things are different on other planets, or maybe the rusty iron got attached during its journey to Earth.
The protein has a fixed chain length, which suggests a living organism as the source, but a living organism would also produce a vast number of other proteins. The fact that it was found 6 mm inside solid rock also argues against a biotic origin. As mentioned, the lithium is likely to be adventitious.
The authors suggest that the protein might have been formed the same way that glycine polymers are formed inorganically on titanium dioxide surfaces. This makes sense, as many chemical reactions that occur in space happen on the surfaces of dust particles in the interstellar medium. They speculate that the iron has a catalytic function, but there is no evidence for this.
Even though it's not evidence of life, it's still scientifically important, as it shows that protein molecules with a potential for enzymatic function can form spontaneously in outer space.
It sounds like the protein is like gelatin, which is one of your Earth proteins formed by hydrolysis of collagen, a protein found in connective tissue and bone. Hydrolysis breaks collagen down into small peptides with a length of about 19 amino acids, though the chunks can be much bigger. Unlike hemolithin, gelatin contains proline and hydroxyproline as well as glycine, plus smaller amounts of other amino acids (glutamate, alanine, and arginine).
So what we have here may be the first discovery of an extraterrestrial form of Jell-O. The authors didn't report finding any Jell-O mold. After this they'll probably go back to the impact site. I bet they'd find one.
mar 03 2020, 7:27 am. edited mar 04 2020, 4:28 am
