
Chemistry of Energetic Materials
G. A. Olah and D. R. Squire
Academic Press, Inc., 1991, 212 pages

|
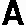 lthough energetic materials have a long
history in construction, demolition, and warfare, there is still a
pressing need for new materials with greater safety and higher energy
content. Understanding the ability of these molecules to store and
rapidly release large quantities of energy could lead to improved
energy storage devices, novel propulsion systems, and new techniques
in materials processing. Chemistry of Energetic Materials
is not a comprehensive textbook on the subject, but a collection
of scientific articles on the chemistry of N-nitramines, which
are an important class of energetic compounds with wide military
application.
lthough energetic materials have a long
history in construction, demolition, and warfare, there is still a
pressing need for new materials with greater safety and higher energy
content. Understanding the ability of these molecules to store and
rapidly release large quantities of energy could lead to improved
energy storage devices, novel propulsion systems, and new techniques
in materials processing. Chemistry of Energetic Materials
is not a comprehensive textbook on the subject, but a collection
of scientific articles on the chemistry of N-nitramines, which
are an important class of energetic compounds with wide military
application.
To a first approximation, the shockwave pressure and specific impulse of a propellant are proportional to the square of the propellant's density. Molecules with cubane and adamantine structures are thus particularly appealing as potential energetic materials not only because their strain energy contributes to energy content, but also because they are unusually dense. R.D. Gilardi and J. Karle discuss the 3-dimensional crystal structure of real and hypothetical materials, such as 1,3,5,7-tetranitrocubane, which has a density comparable to RDX (1.83 vs. 1.81 g/cm3), and hexanitrohexa-aza-adamantane, which is predicted to be 1.5 times as powerful as RDX. Anex et al. describe their studies of the decomposition of 1,3,3-trinitroazetidine, another highly strained molecule related to the familiar 6- and 8-membered cyclic nitramines, using multiphoton infrared photofragmentation spectroscopy. Other chapters investigate the dissociation of haloaromatics using picosecond X-ray diffraction, and computer aided design of monopropellants with properties superior to HMX (1,3,5,7-tetranitro-1,3,5,7-tetraazacyclooctane).
The articles by A.T. Nielsen and G.A. Olah, discussing the chemistry of caged azapolycyclics such as polyazaadamantanes and polyazawurtzitanes, and the more mundane chemistry of nitration reactions, respectively, give an excellent overview of the chemistry of energetic materials. Inorganic chemistry receives short shrift, with only one short chapter on caged lanthanide and alkaline-earth metallacarboranes. The relevance of these molecules to energetic substances is not made clear.
These highly specialized research articles will provide little insight for readers curious about the physical properties of highly energetic materials or the general principles of explosives technology. There is no discussion of peroxides, C-nitro compounds such as trinitrotoluene, or mixtures such as gunpowder, which participates in a very complex series of chemical reactions. However, the articles provide a fascinating glimpse into a specialized branch of the organic chemistry of nitrogen-containing compounds and the exotic instrumentation needed to study their rapid reactions.
