
|
by T Nelson |
Arafat and Polonium
 nly the pathologists who performed the original autopsy on Yasser Arafat
know the true cause of his death on November 11, 2004. Death is, of course,
tragic, even when it happens to someone like Arafat. But the claim that he
was poisoned with polonium-210 is so implausible, and so toxic, that it must
be challenged before it becomes widely accepted as fact.
nly the pathologists who performed the original autopsy on Yasser Arafat
know the true cause of his death on November 11, 2004. Death is, of course,
tragic, even when it happens to someone like Arafat. But the claim that he
was poisoned with polonium-210 is so implausible, and so toxic, that it must
be challenged before it becomes widely accepted as fact.
The pathologists withheld the COD from the public “for privacy reasons,” which means the relatives did not want it disclosed. The public consensus was that Arafat had died of AIDS. If so, it's obvious why the Palestinians would refuse, and it's also obvious why the polonium theory would appeal to them. Po-210 can only be made in a nuclear reactor, and Israel has lots of them. Palestinians are, of course, eaten up with hatred for Israel, but perhaps more importantly there are many others, especially in Europe, who will also celebrate this theory as a way of bashing Israel. So it's necessary to debunk these claims early.
Also, it gives me a chance to teach people some nuclear physics.
Po-210 decay chain and detection limits

Caution Radioactive Material sign
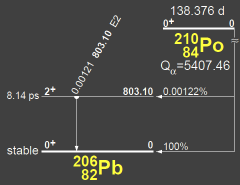
Po-210 is an alpha emitter with a half-life 138.376 days, or 0.3789 years. The decay chain, shown above [1], is very simple: emission of an alpha converts Po-210 to Pb-206, one of the four stable isotopes of lead. (There are 31 other isotopes of lead that are unstable—that is, radioactive.) About 24.1% of natural lead is Pb-206, which means it is impossible to detect polonium by measuring its decay products, because there is lots of lead in the human body.
Nine years is roughly 23.7559 half-lives, which means 7.058×10-8, or about one part in 14 million, of the original amount remains. The quantity of Po-210 we're talking about here is far too small for any chemical or even physical analysis, like mass spectrometry or X-ray fluorescence. So an alpha particle spectrometer would be necessary to identify and measure the isotope. The question is: is it possible to definitively establish a toxic level of Po-210 after 23.7559 half lives? The answer depends on two factors: the sensitivity, or counting efficiency, of the detector, and the background level.
A typical alpha spectrometer is the Canberra Model 8401, which has a counting efficiency of 25%. The background is set by the detector noise or the natural abundance in the sample, whichever is greater. Detector noise is quoted in their sales brochure as 1 count/hour, or 0.0167 counts per minute (measured for Am-241).

Britain's Daily Mail has its own unit for measuring radiation exposure: the Curry Equivalent Dose, or CED
According to the Institut de Radioprotection et de Sûreté Nucléaire (IRSN)[2], the human body contains 30 Bq or 0.18 pg of Po-210, most of which comes from cigarette smoke. However, after 23 half-lives, Po from cigarette smoke would be undetectable. Thus, the detection limit is set by the instrument noise, which is 0.0167 cpm, or by the amount in the surrounding soil and water, which they would also have to measure to make sure it had not entered after burial.
A lethal dose (LD50, the amount that has a 50-50 chance of killing a person) of Po-210 is said to be around 1 microgram, or roughly 0.16 GBq. Pure Po-210 has a specific activity of 1.66×1014 Bq/gram [4], or 4486.5 Ci/gram. Therefore, 1 μg = 4.5 mCi = 9,960,000,000 DPM, which is pretty much.
After 23.7559 half lives, the lethal dose of 9.96 billion DPM would have decayed to 703.1 DPM or 175.8 measurable cpm. This is the amount that would be in his entire body, and it is a very, very small amount; for comparison, the human body contains 264,000 dpm of potassium-40, or 375 times more, entirely from natural sources.
But there's a problem. To measure this amount, they would have to ash his entire body. That's not going to happen. It is known that before his death, Arafat lost about 1/3 of his body weight. Let's suppose his original weight was 165 pounds. His final weight would be 110 pounds. If the polonium was evenly distributed, each gram of his body would contain:
110 x 1000 /2.2 = 50,000 grams 1 gram = 175.8 cpm / 50,000 = 0.0035 counts/minute
This is well below the instrument noise level of 0.0167 cpm. They would have to collect and ash 284 grams to get one count per minute. That's not as easy as you might think—these instruments aren't designed to handle large clumps of stuff, so the sample would have to undergo a heroically complicated enrichment process.
Then there is the question about whether Arafat had been embalmed. If so, much of the polonium would have been flushed out because of its high affinity for red blood cells. Polonium is chemically similar to selenium, and 28-46% is stored in the muscle after long term exposure. But after acute ingestion, the vast majority would be in the red blood cells or the blood plasma. Later, it slowly goes to the liver, spleen, and kidneys. But a significant part of it would remain in the blood, and would be lost.
Also, since the bioavailability of Po-210 is low, it means that a person would have to ingest ten LD50s, or 1-3 GBq, to acquire a lethal dose. The dose, given as polonium nitrate, would therefore be very high in his mouth and throat. This may explain why Alexander Litvinenko, who was poisoned with it by the KGB, had been unable to eat for 18 days due to throat inflammation.
Of course, whoever did it might not have wanted to take any chances, and could have given him a hundred or a thousand times the LD50 instead of ten. So what we really need to know is how many counts per minute the labs detected. It's unlikely that they or Arafat's relatives will ever tell us that.
Pathology of polonium poisoning
But we have ways. According to Harrison et al. [3], death from Po-210 poisoning occurs a few weeks after ingestion, with bone marrow failure an important component. About 0.1-0.3 GBq absorbed in the blood is fatal within 1 month. Since its bioavailability is low, as mentioned above, this means that a person would have to ingest 1-3 GBq.
Alexander Litvinenko experienced vomiting, irregular heartbeat, kidney problems, and a loss of white blood cells, and, most significantly, total loss of his hair within a week. These symptoms, especially sudden hair loss, are classic symptoms of poisoning (such as from chemotherapy) or radiation poisoning. They are not subtle and are difficult to miss. The accumulation in red blood cells, and loss of white blood cells, is well known and easy to measure. The French pathologists had to know the cause of death back in 2004. They refused to release it for privacy reasons—in other words, acceding to the wishes of the next of kin. But it was obvious even then that the Palestinians would try to make a blood libel out of it. Considering the political implications, the pathologists should have insisted on revealing the truth as a condition of their involvement. But if they were honest they also would have said that, after all this time, it was utterly pointless to test for polonium.
References
[1] Firestone RB, Table of Isotopes, 8th ed., 1998, Wiley-Interscience
[2]
http://www.irsn.fr/EN/news/Pages/20061128_impact_of_polonium-210_on_human_health.aspx
[3] J Radiol Prot. 2007 Mar;27(1):17-40. Polonium-210 as a poison.
Harrison J, Leggett R, Lloyd D, Phipps A, Scott B.
[4] hps.org/documents/po210factsheet.pdf
