 he Montreal Protocol, ratified in 1987, forced the industrialized world to switch
from chlorofluorocarbons (CFCs) to hydrofluorocarbons (HFCs) on the theory that CFCs
break down the ozone layer.
he Montreal Protocol, ratified in 1987, forced the industrialized world to switch
from chlorofluorocarbons (CFCs) to hydrofluorocarbons (HFCs) on the theory that CFCs
break down the ozone layer.
Both the formation and depletion of the ozone layer depend on ultraviolet light from the Sun. The theory was that UV splits chlorine atoms from the CFCs. The CFCs sat around all winter, moving into position, waiting ... and then just as the Sun returned, the chlorine radicals chewed up the ozone as it was being formed, producing a brief downward spike in ozone at the start of the Antarctic summer. This is the famous ozone hole.
The actual measurements look very peculiar, which means there's more going on than just a simple chain of free-radical reactions. But NASA and the climatologists were confident that the mystery was solved. As with the AGW debate, most agree that it could theoretically happen; the debate is over how big the effect is and how important it is.
Global Warming Potential (GWP) and Ozone Depletion Potential (ODP)
| Refrigerant | Chemical name | GWP | ODP |
|---|---|---|---|
| HFO-1234yf | 2,3,3,3-tetrafluoropropene | 4 | 0.00 |
| HFO-1234ze | 1,3,3,3-tetrafluoropropene | 6 | 0.00 |
| HFC-134a (R-134a) | 1,1,1,2-tetrafluoroethane | 1430 | 0.00 |
| R-407c | (mixture) | 1774 | 0.00 |
| HCFC-22 | chlorodifluoromethane | 1810 | 0.05 |
| R-410a | (mixture) | 2088 | 0.00 |
| HFC-125 | pentafluoroethane | 3500 | 0.00 |
| HFC-143a | 1,1,1-trifluoroethane | 4470 | 0.00 |
| CFC-11 | trichlorofluoromethane | 4750 | 1.00 |
| CFC-12 | dichlorodifluoromethane | 10900 | 1.00 |
Sources:
Linde,
Engineering Toolbox.
Some values from other sources differ.
GWP is based on carbon dioxide = 1. ODP is based on CFC-12 = 1.
The average GWP of HFCs, weighted by usage, is about 1600.
If viewing on a cell phone, drag table left or right to scroll.
Common refrigerants
Four popular HFCs in use today as refrigerants are R-410a, R-407c, R-143a, and R-134a. The average GWP of the HFCs currently in use, weighted by usage, is about 1600. Enviros are claiming that eliminating these so-called high-GWP HFCs will prevent up to 0.5°C of warming by 2100. Due to the huge variability in the predictions of the various models, this could be anywhere from 8 to 100% of what the models predict. What is remarkable is that absolutely nobody seems to have noticed any of this until the patents ran out.
But never fear: Dupont and Honeywell are ready to save the day with a new class of unsaturated hydrofluoroolefins, namely HFO-1234yf (a.k.a. R-1234yf) and HFO-1234ze. Due in part to a massive lobbying effort, the leading contender for a replacement is HFO-1234yf, a.k.a. 2,3,3,3-tetrafluoropropene.
The companies are well prepared: Dupont, Honeywell, Mexichem, Arkema, Nippon Oil, Daikin, and a few other foreign companies have a total of over 200 patents on production processes for HFO-1234yf as well as patents for blends, equipment, and application. Most of these patents were filed between 2007 and 2012, so the new molecule will be well protected until at least 2032.
Well, corporations will be corporations. But by getting HCFCs banned to protect against global warming, the EPA can convince people that, indeed, there is such a thing as global warming. Then, if and when global warming is proved not to exist, they can take credit for having fixed it. But wait ... maybe they fixed it already, purely by accident.
Why would HFCs and CFCs cause global warming?
Most articles merely say that HFCs cause global warming because they possess a high GWP. This is a circular argument, because GWP simply means global warming potential.
The real explanation is that they absorb thermal (mid-)infrared radiation at wavelengths that don't overlap with carbon dioxide or water. The infrared spectra of HFC-125 and HFC-143a have three bands in the mid-infrared which have little overlap with carbon dioxide (CO2):
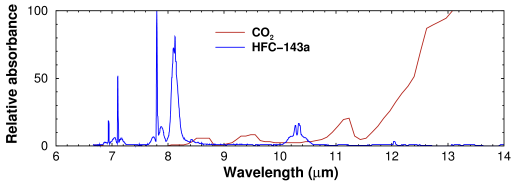
Comparison of mid-infrared absorption maxima for HFC-143a and carbon dioxide. HFC-143a absorbance calculated from absorption cross section at 800 hPa air and 293K; data from Di Lonardo and Masciarelli, J Quant Spec Rad Trans 66, 129–142 (2000). Spectra have been scaled to 100% of maximum absorbance and were measured under different conditions, so the y axis values are not quantitatively comparable.
In spite of what you may have heard, not all scientists are concerned about CO2. One paper by Q.B. Lu [1], for instance, says that CFCs could be wholly responsible for whatever global warming there is. This theory is based on Dr Lu's studies of cosmic ray-induced ozone depletion. CFCs correlate with warming, but CO2 doesn't. Is it really only a coincidence that the ‘pause’ began a few years after CFCs were banned?
Whether you believe it is depends mainly on whether you are convinced that the basic theory is sound. And that seems to depend on whether you're a liberal or a conservative.
Are HFOs dangerous?
Two side issues are affordability and safety. Some enviros are saying that poor countries should have the right to manufacture it without paying royalties, as they're increasingly allowed to do now for drugs. This is a red herring that obscures the real question of whether the new chemical, or any change at all, is desirable.
According to the Daily Mail it's not. They reported that Mercedes Benz refuses to use R-1234yf because it is flammable and highly toxic. Daimler, the parent company of Mercedes Benz, said in 2013 that it caused engine fires when the refrigerant line was cut in a hot engine during a crash. The Daily Mail showed photographs of the burning engine, which they described as a 'fireball' and said that it produces hydrogen fluoride (HF) gas. Daimler recalled every car that contained the new refrigerant.
In the old days we used HF to dissolve proteins for chemical analysis. We stopped because it is deadly poison. If it gets on you, you will lose whatever part of your body it touches, and you won't know it's happening until it's too late. The Daily Mail quotes Professor Andreas Kornath of the University of Munich as saying this makes HFO-1234yf unsafe.
The US EPA correctly points out that HFO-1234yf is less flammable than methane and acetylene and has a higher ignition energy. They claim that HFO-1234yf will remain the only viable low-GWP vehicle air conditioner refrigerant for many years. Even if ignited, they say, HFO-1234yf burns only weakly, and they claim that Daimler's test conditions were unrealistic and they undercharged the AC system. But the danger is not its flammability—it's the HF and carbonyl fluoride that get released during pyrolysis.
Honeywell downplays the risk, saying that temperatures of 700°C are needed to produce HF. Their position is that the risk is comparable to other refrigerants, and that since HF is water soluble it would be washed away by water used to extinguish the fire. Of course, by that time, you've already been exposed.
Teflon pyrolysis
Of relevance is the thinking that the dangers of pyrolysis of Teflon may not be due to HF, but to production of fluoroolefins. Birds are particularly susceptible to fumes from overheated Teflon (PTFE) and related polymers due to their efficient respiratory system. PTFE pyrolysis products include fluoroolefins such as tetrafluoroethylene, hexafluoropropylene, and perfluoroisobutylene. The most toxic product, COF2, is produced above 480°C. Polymers containing hydrogen, such as Viton A, also produce ‘significant’ amounts of HF. I could find no toxicological studies of HFO-1234yf on birds.
And so, just as the old patent is about to expire, the EPA wants to ban the safe ones and force us to switch to a new one which one highly respected corporation says produces toxins in a crash and another one says it doesn't. We're also seeing articles from libs ominously telling us we don't really need A/C at all, and it's fun and romantic to sit in a puddle of sweat all day as we used to do back in the old days.
And we're seeing hints of how the need for a third generation of refrigerants will manifest itself. Anything containing fluorine is hazardous, they'll say. Or maybe birds are dropping from the skies. Gazing into my silicon dioxide crystal ball, I predict it will happen ... oh, round about the time the patent runs out.
The Pause
But the more I think about it, the more convinced I am that if there was man-made warming, it ended—purely by accident—when they got rid of CFCs.
It's generally agreed that there has been a ‘pause’ in global warming for the past 15 to 20 years. Although CFC-12 has a lifetime of 120 years, far too long to see any effect yet on ozone levels, CFCs have stopped accumulating in the troposphere since their production was banned in 1990. In 2000, ten years after their production was banned, tropospheric CFC concentrations started declining. This matches almost to the year the start of the pause. Maybe that's not a coincidence ....
CO2 is 0.04% of the atmosphere. If CFC-12 has a GWP of 10900, it would have the same effect as CO2 at 36.7 parts per billion. The actual midyear tropospheric concentration of CFC-12 in the northern hemisphere, according to the Oak Ridge National Laboratory[2], is below this: 0.521 ppb.
However, we know that the effects of so-called greenhouse gases are highly nonlinear. They are logarithmic, which means that the lowest concentrations produce a disproportionately large effect. Going from 0.0 to 0.01% has a vastly bigger effect than going from 0.04% to 0.05%. Remember that the total warming is not 1 or 2 degrees, but 33°; thus this 0.521 ppb of CFC-12 could easily be enough to cause the 0.5° or so of warming we've seen in the last century. Even if the relationship were linear, we can see that CFCs could cause this amount of warming: 0.521 ppb CFCs / 36.7 ppb CFCs = 0.0142 is close to the observed warming, or 0.5° / 33° = 0.0152.[3]
Of course these are just BOTECs (back-of-the-envelope calculations). But suppose we accept the warmers' estimate that going from HFCs to HFO-1234yf will prevent 0.5° of warming. Based on their weighted GWPs, that means that going from CFCs (assumed to be entirely CFC-11) to HFCs must have saved 0.5 × (4750 − 1600) / (1600 − 4) or 0.9868° of warming already. This more than accounts for the pause. If we calculate it as CFC-12, it works out to 0.5 × (10900 − 1600) / (1600 − 4) or 2.914° of warming. If we take the warmers at their word, the ozone layer has cured global warming!
1. Int J Modern Physics B 27(17), 2013, 1350073 Cosmic-ray-driven reaction and greenhouse effect of halogenated molecules: culprits for atmospheric ozone depletion and global climate change (trigger warning: paywall) (non-technical summary here) arXiv
2. Bullister, J.L. 2015. Atmospheric Histories (1765-2015) for CFC-11, CFC-12, CFC-113, CCl4, SF6 and N2O. NDP-095 (2015). Carbon Dioxide Information Analysis Center, Oak Ridge National Laboratory, US Department of Energy, Oak Ridge, Tennessee. doi: 10.3334/CDIAC/otg.CFC_ATM_Hist_2015. Link. PDF
3. Based on the IPCC's own numbers for radiative forcing: CFC-11 = 0.07 Wm−2, CFC-12 = 0.17 Wm−2; total of 0.24 is 14.46% of the 1.66 Wm−2 from 379 ppm of CO2). If all factors were equal, this would work out to 0.1446 × 33 = 4.77° from CFCs. Interestingly, 4.77 × 1600 / 10900 = 0.7. Maybe something like this is where the warmers got the 0.5° figure for HFCs.
Revised and updated Aug 01 2016; last updated Aug 14, 2016, 5:16 am
